The Nocebo Effect
You’ve heard of the placebo effect – the positive effect a suggestion can have on others. Now for the nocebo effect – the negative effect a suggestion can have on others.

By Ramesh Mehay
GP Trainer (Yorkshire & the Humber)
A lot of the side-effects connected to prescribed medication could be down to the “nocebo effect”. In other words, some patients will experience the side-effects they are informed of, even if the drug is a placebo. And this is backed up by a evidence.
Let's start by looking at statins
These are one of the most commonly prescribed drugs in the UK. Did you know that up to a fifth (20%) of individuals stop taking them because of side-effects? Things like tiredness, muscle mass pains, joint discomfort, nausea and vomiting. Yet the medical research says the incidence of side-effects is much lower. So why is there this variance between what patients perceive and what the research says is real?
There was an interesting study led by Frances Wood and Dr James Howard at Imperial College London published in the New England Journal of Medicine. What happened? They got 60 patients who had been on statins but stopped taking them because of side-effects. They persuaded them to restart treatment. EXCEPT this time they gave them four bottles of atorvastatin, four of identical-looking placebo pills, and four empty bottles. They prescribed them randomly over a year. Here’s the exciting bit: 90% of the negative symptoms recorded in their diaries was when they took the placebo! There were 71 times when people stopped taking them altogether because they said the symptoms were too overwhelming – 40 during the statin months and 31 during the placebo months!
The research convincingly showed that while individuals had different symptoms that they might associate to statin use, symptom scores were no higher throughout periods of statin use compared to the duration of no statins. This provides some proof that the nocebo effect plays an important role in negative side-effects credited to statins.
Now let's look at antidepressants
Of course, the study above does not provide a clear answer on how this exactly applies in a wide range of different types of patient. But thinking logically, is it possible the nocebo effect works across all drugs? Why should it just be for statins?
For example, with antidepressants: should we tell patients that when initiating the drug, that they may get worse before getting better? Are we not putting that thought in the patient’s mind? Aren’t we at danger of sowing that idea which the patient then goes on to fertilise and water over the next few weeks?
The research says that about 15% should genuinely feel worse first before getting better. But studies suggest 80% of people stop antidepressants within a month (others say 40%). Both are incredible numbers! My question to you is this: is it possible that a lot of patients stop antidepressants within a month because they experience what we have told them. And this is the crucial point: many of them might be experiencing the nocebo effect in response to what we’ve said rather than real symptoms.
The words we use as doctors have a massive impact on patients – we know that already – numerous studies provide evidence for this. This has changed the way we practise and perfect our communication skills with patients. But often the learning point is a rephrasing of how we say things. I am not suggesting a rephrasing or paraphrasing here. I am suggesting a complete omission!
Should we tell patients when initiating an antidepressant, that they may get worse before getting better? Are we not putting that thought in the patient’s mind? Aren’t we at danger of sowing that idea which the patient then goes on to fertilise and water over the next few weeks?
Why I am suggesting a complete omission: imagine saying things in a more balanced way like “A very small number might get…..“. You will get several patients who will say “I bet I will be one of them”. And you know as well as I do at this point that their self-fulfilling prophecy will be fulfilled.
If we STOPPED saying things like “you might feel worse before you get better”, perhaps that might prevent the nocebo effect from taking hold and thus an improvement in tolerability scores and concordance? I understand why we, as doctors, inform patients about these things. We want patients to be in the know, to anticipate things and to try and persevere and continue. But does it have the opposite effect? And when that patient stops taking that antidepressant – which might have otherwise done a lot of good – does that then become a disservice to our patients. We all know how depression is a significant mental illness which, if left untreated or unmanaged, can cause a tremendous amount of distress for a patient, their family and friends.
And finally, time now to move from subjectivity to objectivity – let’s close with some figures.
- The reality is that 60% of people respond by about 2 months to antidepressants with about a 50% reduction in symptoms.
- So, out of 10 people, 6 will be better by 2 months. 4 may not.
- In terms of “getting worse before getting better” – the real figures is more like 2 out of 10 will experience this.
- But if we highlight it, the nocebo effect kicks in. This number climbs from 2 to 4, 5, or even 6.
- So, why should all 10 patients be warned of this? Why put the other 8 at risk of the nocebo effect when the reality is that the real symptoms will only be experienced by 2?
- Why not work with the 2 – if and when they get it?
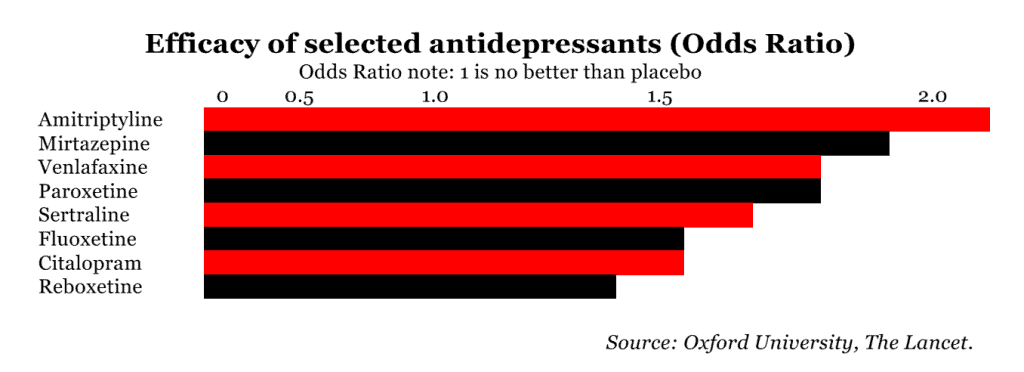
An interesting study in 2018...
Systematic review and meta-analysis of double-blind RCTs assessing antidepressants for depressed adults. 522 studies. 116477 patients in total. Effectiveness defined as 50% or more improvement in symptoms plus good tolerability.
Results
- People were equally likely to stop taking the antidepressant as the placebo!!! [except for clomipramine – which was more intolerable). Again, does the nocebo effect have a lot to answer for?
- Fluoxetine better tolerated than the rest.
- Other good ones – escitalopram, paroxetine, sertraline, mirtazipine, agomelatine (an atypical antidepressant).

Nocebo effects are very important. I spend a lot of my time in the pain clinic undoing vivid mental images that patients have of “crumbling spines”, “discs like wafers with bone grinding on bone” and lots of other needlessly negative notions they have collected during their journey through the healthcare system. Degenerate is a word used by radiologists, but one which is likely to make patients freak out.
The old Second World War posters said: “Careless talk costs lives”. This is true in medicine, where patients stop living their lives for fear of destroying their spines/hips/necks, etc.
Some of the most florid descriptions of non-existent harm come from spinal manipulators who seem to want patients to become therapist-dependant, which is the exact opposite of what we are trying to achieve with our efforts to activate patients and get them contributing to their own health.
Thanks for this Gareth. Yes, words like “crumbling spine” are simply awful. Imagine what a non-medical lay person must think. Would you buy a house off me if I said it was “crumbling” in places? And for the same reasons, I don’t like the words “wear and tear”. Would you by a belt or item of clothing if i said it was torn in places? And the way we medicalise MILD degenerative changes. Yet, we never say to a patient that they have “degenerative skin” when it simply plain old wrinkles – which is part of a natural progression of life!